How one little girl helped researchers stump the mumps
Chipmunk cheeks may sound cute, but swollen testicles, seizures and encephalitis are not nearly so charming. Celebrate 50 years of the mumps vaccine, which has largely eliminated this once universal child illness.

Mumps quarantine sign, from sometime after 1909
Illustrations often comically portray the famous calling card of mumps—swollen jaw and cheeks. Because so many of us are now protected by the mumps vaccine, we never experienced those cheeks, nor do we suffer the lasting indignity of parental photographs of our puffy faces. Mumps cheeks became an artifact of the American past—the perfect subject for Norman Rockwell illustrations of a family’s fishing vacation canceled at the appearance of the dreaded swelling.

Two prints by Norman Rockwell, around 1972. Merck Research Laboratories commissioned the works to celebrate vaccination and the changing American experience of childhood disease. (Courtesy National Museum of American History)
Most people who contract the disease recover fully. However, beyond those chipmunk cheeks, mumps can have serious (and sometimes permanent) complications that are not so comical: inflammation of the testicles and ovaries, encephalitis (swelling of the brain), meningitis (infection of the tissue around the brain and spinal cord), seizures, and deafness.
Fifty years ago, Americans began to forgo chipmunk cheeks and swollen testicles, when a hard-won mumps vaccine became available.
In the 1960s, Dr. Maurice Hilleman, the chief virologist at Merck Research Laboratories, set his mind to creating a mumps vaccine. He did so knowing that although mumps was a universal disease of childhood, many parents underestimated its danger. According to Dr. Paul Offit’s book Vaccinated, during the 1960s “mumps virus infected a million people in the United States every year.” Scientists had been working to develop a safe and effective vaccine against mumps without success: the vaccines caused either an unacceptably high level of mumps symptoms, or an unacceptably low level of immunity. Dr. Hilleman suspected he needed to start anew with a different strain of the virus—and he got his chance when his daughter Jeryl Lynn awoke him one night to show him her swollen cheeks. That night Hilleman swabbed her throat to collect a sample of the virus, and then drove the sample to his lab.

Jeryl Lynn Hilleman displays her swollen cheeks in 1963
In the ensuing days, Hilleman set to work hoping to weaken, or attenuate, the virus so that it would not cause mumps, but could still provoke immunity. He accomplished this by introducing the virus to generations of chicken embryo cells. With each successive generation, the virus changed: it became better and better at attacking the chick cells, and consequently became worse and worse at attacking human cells.
Human testing of Hilleman’s officially designated “Jeryl Lynn Strain” vaccine proved that it achieved that delicate balance of producing strong immunity without causing the disease in vaccinated children. Although Jeryl Lynn could not benefit directly from the vaccine that bears her name, her younger sister did. In 1966 Kirsten Jeanne Hilleman was immunized with the investigational vaccine. In early 1968 doctors began administering the Jeryl Lynn Strain mumps vaccine to the American public.
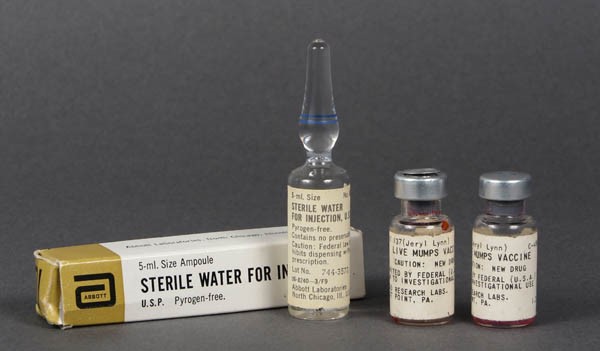
The vials of investigational vaccine used to immunize Kirsten Jeanne Hilleman against mumps.

A 1966 photograph that accompanied Merck’s press release about the successful vaccine depicted Jeryl Lynn comforting the crying Kirsten as the younger girl received her vaccination with the Jeryl Lynn Strain from Dr. Robert Weibel.
Recently, the American History Museum acquired an intriguing object that documents how—for 50 years—we have all relied on Jeryl Lynn’s throat swab that fateful night: a shiny stainless-steel 15-liter canister. The canister is one of five in which the entire supply of the frozen Jeryl Lynne virus “seed stock” was kept safe and pure. For years this canister had safeguarded stock that could produce 3.26 billion doses of vaccine . . . that is, until its green neoprene stopper began to fail.
In 2015 Merck scientists embarked upon an anxious task: they had to thaw and transfer the precious seed stock to new containers, all while keeping it untainted. If the stock had lost its potency, or became contaminated during the transfer, it would take five to seven years to get new stock into production. This could cause a gap in the vaccine supply.

The stainless steel container that once housed the virus seed stock (courtesy National Museum of American History)
The transfer was successful, and the Jeryl Lynn stock now resides within a more secure type of container. The old mumps canister has come to live at the Smithsonian. Like the millions of Americans protected by the strain it once sheltered, the canister is now mumps-free.
This post by Rachel Anderson, a research and project assistant in the Division of Medicine and Science was originally published by the American history Museum blog, O Say Can You See?
Posted: 20 December 2018
-
Categories:
American History Museum , History and Culture , Science and Nature





